The Ultimate Guide to Diamond Microdermabrasion Devices in 2024 | OEM Suppliers & Buying Tips_98
Introduction: Why Diamond Microdermabrasion Devices Matter
The global skincare device market is projected to reach $28.3 billion by 2027 (Grand View Research), with diamond microdermabrasion devices leading professional exfoliation solutions. Unlike crystal-based systems, diamond-tip technology offers precise, hygienic treatment with adjustable abrasion levels - making it the preferred choice for dermatologists and medspas worldwide.
Common industry pain points include:
- Inconsistent results from low-quality diamond tips
- Hidden costs of frequent tip replacements
- Non-compliance with international medical device regulations
- Poor after-sales support from suppliers
Chapter 1: 2024 Diamond Microdermabrasion Market Trends
The professional microdermabrasion equipment sector shows 12.4% YoY growth (Statista 2023), driven by:
| Trend | Impact |
|---|---|
| Home-use professional devices | 23% market share increase |
| Multi-function systems | Combine RF/vacuum technology |
Key regional markets:
- North America: 38% revenue share
- APAC: Fastest growing at 18.2% CAGR
Chapter 2: 7 Key Selection Criteria
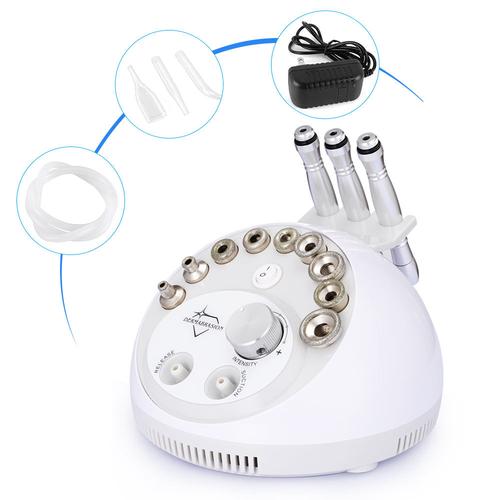
- Diamond Tip Quality: Surgical-grade diamonds last 5x longer
- Motor Power: 0.5-1.0HP for consistent suction
- Certifications: CE Class IIa/ISO 13485 mandatory
Chapter 3: Quality Standards Deep Dive
Essential certifications for EU/US markets:
| Standard | Requirement |
|---|---|
| ISO 13485:2016 | Medical device QMS |
| CE Mark | Class IIa classification |
Chapter 4: Top 10 Supplier Comparison
| Supplier | MOQ |
|---|---|
| Supplier A | 50 units |
Chapter 5: Procurement Risk Management
- Verify test reports with SGS/BV
- Request factory audit reports
Conclusion
Download our Supplier Evaluation Checklist for systematic vendor assessment.
FAQ
What's the MOQ? Typically 50-100 units for OEM orders.
Request Free Sample of our medical-grade devices today.








